Enhancing the review, supervision and maintenance of medical devices is a timeless topic
The major problems caused by the popularization of staplers have sounded the alarm for all medical devices. For all medical devices that are ready to go on the market or are in use, the management of the following aspects needs to be strengthened to reduce the occurrence of injuries:
1. Strengthen the review of medical devices and kill dangerous devices in the cradle
The stapler was previously classified as a Class I device, and it was the loose review system that led to the entry of staplers of uneven quality into the market. Under the conditions that the review cost allows and is reasonable, strengthening the review and safety assessment of medical devices is the most effective measure to reduce risks.
2. Give medical equipment an "ID card"-eliminate high-risk, second-hand equipment
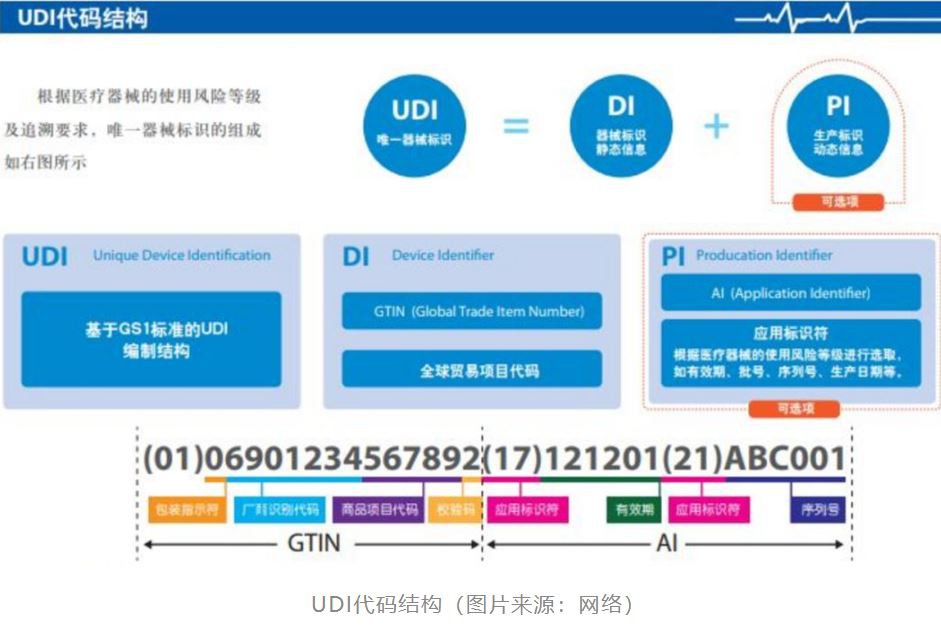
The US FDA has established a "special medical device identification system"-Unique Device Identification (UDI). The identification given to medical devices throughout their life cycle is their unique "identity in the product supply chain" certificate". Medical devices have entered the era of traceability and supervision of the whole process from production, processing to circulation, distribution, and use.
The system is gradually being accepted worldwide. The United States and the European Union have already implemented it, and Turkey, South Korea, India, etc. have put UDI regulations on the agenda.
Recently, the State Food and Drug Administration of China held a seminar on the pilot work of the unique identification system for medical devices, and plans to establish a unified traceability system using unique device identification (UDI) across the country. Completely eliminate high-risk, second-hand and substandard medical devices.
3. After-sales training and maintenance are important links to ensure the usability and safety of equipment
The functions of modern medical equipment are becoming more and more abundant, the level of science and technology is becoming more and more sophisticated, the degree of precision is getting higher and higher, the complexity of equipment operation and the difficulty of maintenance are becoming more and more Medical equipment malfunctions, leading to adverse events that endanger patient safety.
Hospitals generally report that many manufacturers are unable to provide good after-sales services, especially for "manufacturers' on-site maintenance", "after-sales training for hospitals", "manufacturer hotline service" and other aspects of satisfaction.
The stapler is a device used in surgery to replace manual suture. The working principle is to use titanium nails to break or anastomose tissues, similar to a stapler. The mechanized operation method makes surgical sutures more efficient, and makes many difficult and complicated operations easier, and greatly reduces the incidence of surgical complications.
After more than a century of development, the types of staplers are diverse and their popularity is wide. North American and European medical equipment giants (Johnson & Johnson, Medtronic, etc.) lead the global market for staplers. In the late 1970s, China began to independently develop staplers. At present, domestic stapler manufacturers are mainly distributed in Jiangsu, Shanghai and Beijing. The domestic open stapler can be replaced by imports to a certain extent, and there is still much room for improvement in the endoscopic stapler.
However, the looser review and supervision environment has led to serious consequences, causing tens of thousands of failures, nearly 10,000 serious injuries and hundreds of deaths in the past few years. The FDA issued a statement on April 23, proposing to reclassify the surgical stapler as a high-risk medical device and require pre-market review and performance and effect research on the device. This has also sounded the alarm for other booming medical devices, ensuring product safety, realizing full-process supervision, and ensuring after-sales training and maintenance services are necessary conditions to ensure the long-term development of enterprises and products.
Surgaid Medical (Xiamen) Co., Ltd
Surgaid Medical (Xiamen) Co., Ltd has been committed to surgical product development ,production and marketing for 10 more years . In particular, minimally invasive surgical products. Minimally invasive surgery is a surgical direction of development. Such as Disposable Laparoscopic Trocar, Linear Cutter Stapler, Circular Stapler, PPH, and so on.
Manufacturing our own Surgical products allows us to have complete control over the design and evolution of our products. We are able to adapt quickly to the constantly evolving healthcare market and offer high quality, single-use, cost-effective solutions that directly compete with global market-leaders.
Surgaid products:
For more product information at https://www.surgaid-medical.com/