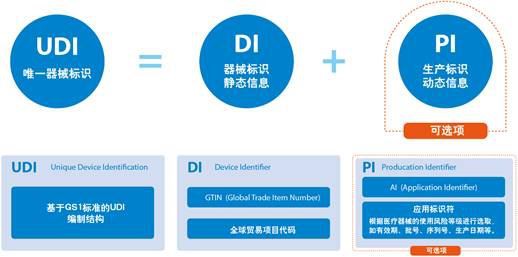
1.What is the unique identification (UDI) for medical devices? What is included?
The unique identification of a medical device refers to a code composed of numbers, letters or symbols attached to the product or packaging of a medical device to uniquely identify the medical device.
The unique identification of medical devices includes product identification and production identification.
2.What is the product identification (DI) in UDI?
The product identification is a unique code identifying the registrant/filer, the model specification and packaging of the medical device.
3.What is the production identification (PI) in UDI?
The production identification is composed of the code of the relevant information of the medical device production process. According to the requirements of supervision and actual application, it can include the medical device serial number, production batch number, production date, expiration date, etc.
4.What product scope does the UDI system rule apply to?
Medical devices sold and used within the territory of the People's Republic of China.
5.Who is responsible for creating and maintaining UDI?
The registrant/filer is responsible for creating and maintaining the unique identification of medical devices in accordance with these rules.
6.Which packaging levels require UDI?
The registrant/filer shall… assign a unique identification data carrier to the smallest sales unit of medical devices and higher-level packaging or medical device products listed in their name.
7.Can UDI be implemented using GS1 standards?
can. According to Article 10 of the UDI System Rules, the code issuing agency shall be a legal entity in China with a complete management system and operation system to ensure the uniqueness of the medical device unique identification created in accordance with its standards and meet the relevant national data security requirements.
GS1 China fully meets this requirement, and the GS1 standard has been successfully implemented in the domestic medical device traceability field for more than ten years.
8.How to apply for GS1 vendor identification code to implement UDI, is there any fee?
Companies can apply for GS1 vendor identification code to implement UDI in the following four ways. For specific fees, please refer to the fee announcement: http://www.gs1cn.org/Business/Barcode/Corppay.aspx
9.How to check whether a company has applied for a manufacturer identification code?
You can inquire according to the manufacturer name or manufacturer address on the homepage of the coding center website:
http://www.gs1cn.org/
10.Can a medical device produced by a domestic enterprise and sold directly in the country (including imported and repackaged in the country) be used as UDI?
First of all, you should confirm that the subject of applying for the GS1 manufacturer code is the "registrant/filer" and that the product barcode meets the requirements of relevant domestic laws and regulations. After that, it can be used after the overseas code is filed in the coding center. For details, see "Notes on the use of overseas registered commodity barcode filing" :
http://glb.ancc.org.cn/WSBA/help.pdf
11.Does GS1 meet UDI regulations in other countries? Can the manufacturer identification code applied for from the China Article Numbering Center be used for export product UDI?
GS1 is a UDI code issuing agency recognized by the United States and the European Union, and it also meets the UDI related regulations that have been issued by countries around the world. The manufacturer identification code assigned by the coding center can be used to compile UDI in compliance with foreign regulations.
Our products:
For more product information at https://www.surgaid-medical.com/
Surgaid will participate in MEDICA online exhibition, please don't miss it.