Everyone knows that one of the most important features of sterile medical devices is sterility.
Among several industrial sterilization methods, ethylene oxide sterilization is widely used in the sterilization of medical devices due to its wide range of application, low cost, and operability.
01. Principle of ethylene oxide sterilization
Ethylene oxide gas is a small molecule chemical substance that is prone to polymerization and alkylation. It is extremely easy to undergo oxidation-reduction reaction with the carboxyl group in life macromolecules, thereby destroying the functional structure of bacteria and achieving the purpose of inhibiting the growth of bacteria.
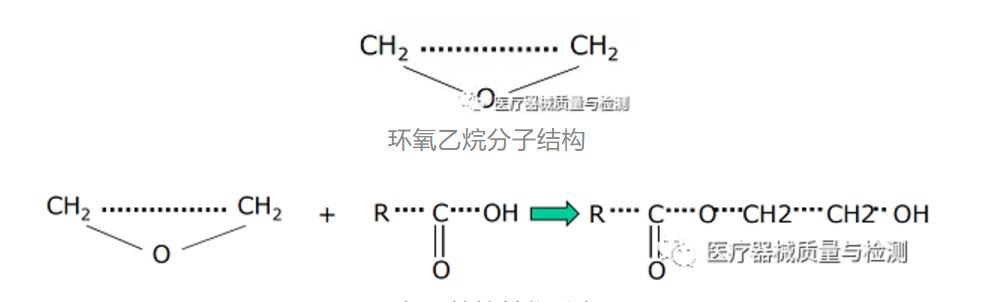
The life macromolecules that undergo alkylation reaction with ethylene oxide are mainly proteins and nucleic acids. Protein is the executor of important life functions of the bacterial membrane system, and nucleic acid is the carrier of genetic information for bacterial reproduction. After these two molecules are alkylated, the high-level structure of life function molecules will be destroyed, thereby blocking the various activities of bacteria and their spores. This is the mechanism of ethylene oxide sterilization.
02. Consideration of product sterilization in the development stage
In recent years, my country's medical device industry has developed rapidly, especially in the field of high-end medical device tools, such as various new minimally invasive devices and various new material devices. In these areas, many companies are in the product development stage. If the final form of the product provided is a sterile device, a good product planning should consider the sterilization method and the requirements of the sterilization method for the performance, structure, material and other aspects of the product at the beginning of the project. So what are the main aspects to consider?
The first is the product material
The key to consider is whether the material reacts with ethylene oxide gas. Generally speaking, accessories products (such as cotton yarn products, non-woven products), plastic products (such as various pipes, catheters, infusion products) and metal products (such as staplers, orthopedic products, cardiovascular products, etc.) Suitable for ethylene oxide sterilization. However, some products have negative effects such as discoloration after ethylene oxide treatment. There are also some instruments with liquid, which may not be suitable for ethylene oxide sterilization.
In addition, the special requirements of materials should be considered, various coatings (such as heart stent drug coatings, orthopedic equipment bioactive coatings, etc.), products with special requirements on temperature and humidity (such as some products have requirements on temperature, such as silica gel) Mat high temperature deformation), at the beginning of design and development, whether it is suitable for ethylene oxide sterilization should be considered and verified. For powdered products, the effects of humidification and negative pressure in the sterilization process should be considered. In this way, the terminal sterilization form of the product is fully considered from the beginning of the R&D planning, which can greatly reduce the setbacks in the enterprise R&D and registration process.
Followed by product structure
Some medical device products have a cavity structure (such as a balloon), and some products have a very long pipeline (such as a protective coil for various guide wires). Because of the difficulty of gas passage and diffusion, these special structures will pose challenges to the sterilization process. It is worth noting that when ordinary infusion products have protective caps and three-way valves, sometimes the product state of the finished product specifies the on-off of the internal passage of the product, which affects the feasibility of the product's gas passage. Therefore, try to consider the delivery status of the product at the beginning of the product design, and try to keep the product cavity open.
Finally, the product packaging method
Not all packaging methods are suitable for ethylene oxide sterilization. For some product packaging that cannot be "breathed" with EO gas, the feasibility of ethylene oxide gas sterilization should be carefully considered. The requirements in this regard can be found in related packaging series standards such as EN868 and ISO11607.
There is also a special state that requires attention:
Your product is provided in a non-sterile state when it leaves the factory, but the sterilization parameters are recommended in the product manual, and the user unit will sterilize it before use. In this case, the company should submit a confirmation report of the recommended sterilization parameters when submitting the registration materials.
Companies should carefully plan the above aspects at the beginning of product development to ensure the smooth progress of your product during the registration and production process.
Surgaid Medical (Xiamen) Co., Ltd
Surgaid Medical (Xiamen) Co., Ltd has been committed to surgical product development ,production and marketing for 10 more years . In particular, minimally invasive surgical products. Minimally invasive surgery is a surgical direction of development. Such as Disposable Laparoscopic Trocar, Linear Cutter Stapler, Circular Stapler, PPH, and so on.
Manufacturing our own Surgical products allows us to have complete control over the design and evolution of our products. We are able to adapt quickly to the constantly evolving healthcare market and offer high quality, single-use, cost-effective solutions that directly compete with global market-leaders.
Surgaid products:
For more product information at https://www.surgaid-medical.com/