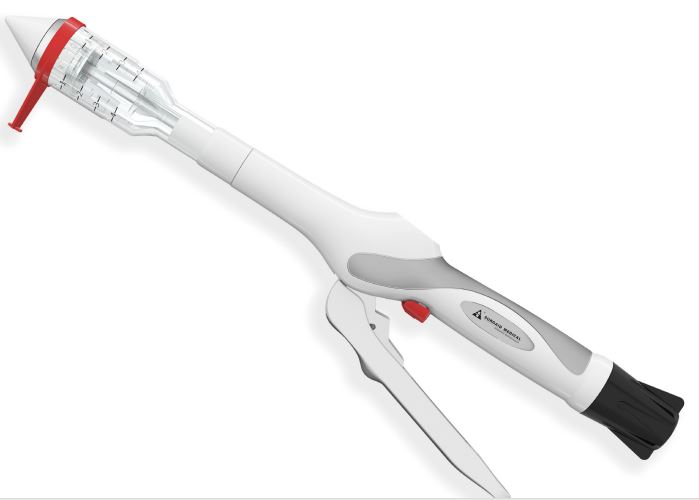
The era of savage growth of the stapler is coming to an end, and the FDA classifies it as a high-risk device category.
Suture is an essential and very important part of surgery.
When it comes to surgical sutures, you may have pictures of doctors stitching up the wound one by one. Indeed, many operations today still require doctors to use superb "sewing" skills to stitch wounds or organ tissues. But sometimes they can also use a medical "stapler"-stapler to make surgical sutures more efficient through mechanized operation, and make many difficult and complicated operations easier, and greatly reduce the incidence of surgical complications.
Although the stapler is convenient, there are always some controversies in terms of safety. In the hit TV series "Surgery Wind and Cloud" in 2017, doctors and patients’ family members had a disagreement on the use of staplers. These issues involved: the doctor’s habits, the doctor’s trust in the instrument, the surgical cost, and the patient’s tolerance, Power and so on. It can be seen that although the stapler is fast and convenient, there are some problems that cannot be ignored.
In recent years, the popularity and barbaric growth of staplers have caused serious consequences, including: tens of thousands of failures, nearly 10,000 serious injuries and hundreds of deaths. In view of this, the FDA issued a statement on April 23 this year, proposing to reclassify surgical staplers as high-risk medical devices, and pre-market review and performance and effectiveness studies of the devices are required. In the future, all surgical tissue binding equipment may be classified as Class II equipment, which means that pre-marketing review and approval pre-market notification are required.
Popularization does not mean safety, "abuse" raises risks
Compared with manual suturing, the stapler is more convenient for surgical operation and can shorten the surgical operation time, and the benefits in the surgical environment are self-evident. Over the past century, the types and functions of staplers have been continuously increased and improved, and sales have also risen sharply. However, these seemingly simple and convenient surgical instruments have "unknowingly" caused tens of thousands of medical accidents. Due to the development of usage habits and the huge interest relationship, people selectively ignore these major safety accidents that have occurred and are still occurring.
Since 1988, surgical staplers have been classified as Class I devices as part of the low-risk, "general purpose manual surgical instruments" specified by the FDA. The loose censorship system has led to stapling devices of uneven quality entering the market and reaching the hands of medical workers. At the same time, device manufacturers have insufficient after-sales training for hospitals, and doctors have different levels, which greatly increases the risk of using such devices.
According to FDA statistics, from January 1, 2011 to March 31, 2018, more than 32,000 malfunctions, more than 9,000 serious injuries, and 366 deaths related to surgical staplers occurred in the equipment.
This includes inability to anastomosis and equipment failure, as well as the deformity of the staples in the wound or the reopening of the wound after the anastomosis. In addition, the report also involved some doctors' operating errors, such as selecting the wrong suture size during the anastomosis procedure.
The U.S. Food and Drug Administration (FDA) issued a statement on April 23, 2019, proposing to reclassify surgical staplers as high-risk medical devices and require pre-market review and performance and effect studies on the devices. In the future, all surgical tissue binding equipment may be classified as Class II equipment, which means that pre-marketing review and approval pre-market notification are required.
Surgaid Medical (Xiamen) Co., Ltd
Surgaid Medical (Xiamen) Co., Ltd
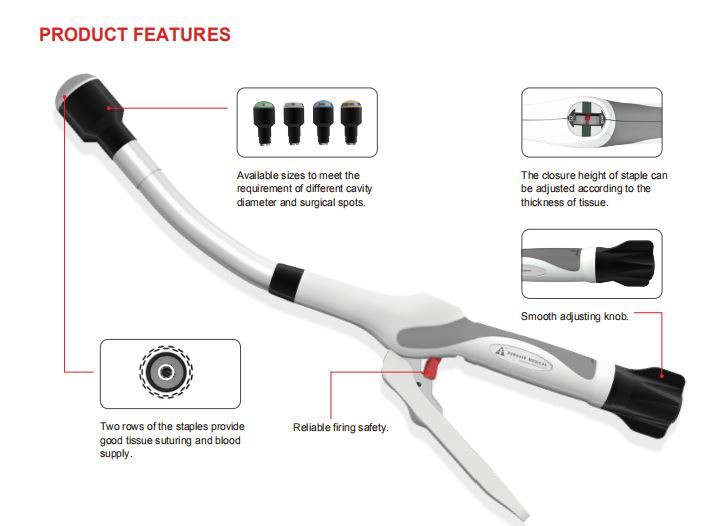
Surgaid Medical (Xiamen) Co., Ltd has been committed to surgical product development ,production and marketing for 10 more years . In particular, minimally invasive surgical products. Minimally invasive surgery is a surgical direction of development. Such as Disposable Laparoscopic Trocar, Linear Cutter Stapler, Circular Stapler, PPH, and so on.
Manufacturing our own Surgical products allows us to have complete control over the design and evolution of our products. We are able to adapt quickly to the constantly evolving healthcare market and offer high quality, single-use, cost-effective solutions that directly compete with global market-leaders.
Surgaid products:
For more product information at https://www.surgaid-medical.com/